AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
 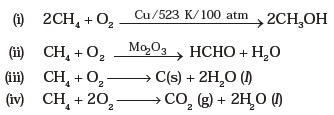
Photoelectron (Photoemission) Spectroscopy (PES)īond Length, Strength, and Magnetic Propertiesġ2 Solids, Liquids, and Intermolecular Forces Percent Composition and Empirical Formulas General Aspects of Chemical Reactions and Equations STEP 4 Review the Knowledge You Need to Score Highĭimensional Analysis-the Factor Label Method Three Approaches to Preparing for the AP Chemistry Exam Questions Frequently Asked About the AP Chemistry Exam This chemical reaction produces smaller hydrocarbons, including alkanes and alkenes.Įthene and other alkenes are unsaturated unsaturated: An unsaturated compound contains at least one double or triple bond.Ĭracking Fuels made from oil mixtures containing large hydrocarbon hydrocarbons: Hydrocarbons are a group of compounds which contain the elements hydrogen and carbon.1 What You Need to Know About the AP Chemistry Examīackground of the Advanced Placement Program In order for it to be useful to us, crude oil is broken down in oil refineries into its component parts, known as fractions, and these can then be used for many different purposes.įractions that are produced by the distillation distillation: Distillation is when we make a liquid evaporate and then condense the vapour back to a purer liquid. Molecule formed in the equation given for cracking in the previous section.Īs shown to the right, thermal cracking is the process of cracking larger molecules by using heat and this case steam. Suggest an equation which illustrates the cracking of decane C10H22 Hydrocarbons are.Ĭracking is an example of a thermal decomposition reaction (in this case of an. These processes break chemical bonds in the molecules, causing thermal decomposition reactions.Ĭomplete the equation for cracking decane C10H22 Update: C10H22 C2H4. Write an equation for the incomplete combustion of decane, C. Write an equation for the thermal cracking of one molecule of C. Nonane is a linear alkane hydrocarbon with the chemical formula C9H20.Ī brief description of the difference between thermal and catalytic cracking of alkanes. Nonane is present in numerous plant oils including olive oils. In the realm of global economics and politics, that statement is hard to dispute.Ĭracking can be classified as a thermal decomposition reaction.Īmong the two long-known allotropic forms of carbon, sp 2-hybridized multilamellar graphite is the thermodynamic stable form at normal temperature and pressure.ĭecane is an alkane hydrocarbon with the chemical formula C 10 H 22. Cracking allows large hydrocarbon molecules to be broken down into smaller, more useful. The Chemistry of the Cracking of Hydrocarbons including the Conditions and Catalyst - Thermal. GROUNDWATER: n-Decane was detected in thermal spring spa water. The substance defats the skin, which may cause dryness or cracking. 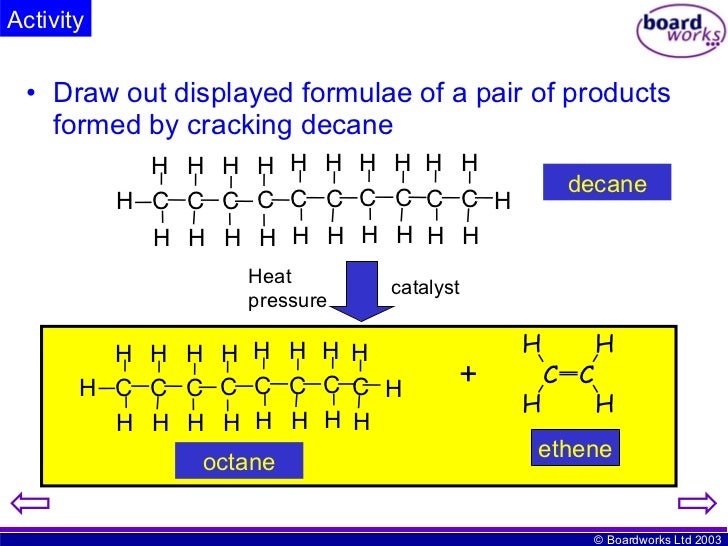
Share this Share through email Share through twitter Share through linkedin Share through facebook Share through pinterest Not the right resource See other similar resources 5.00 BUY NOW Save for later Not quite what you were looking for Search by keyword to find the right resource. BUY NOW Save for later 5.00 BUY NOW Save for later Last updated 3 September 2018 GJHeducations Shop 4.496733668341707 755 reviews A Science teacher by trade, Ive also been known to be found teaching Maths and PE However, strange as it may seem, my real love is designing resources that can be used by other teachers to maximise the experience of the students. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
